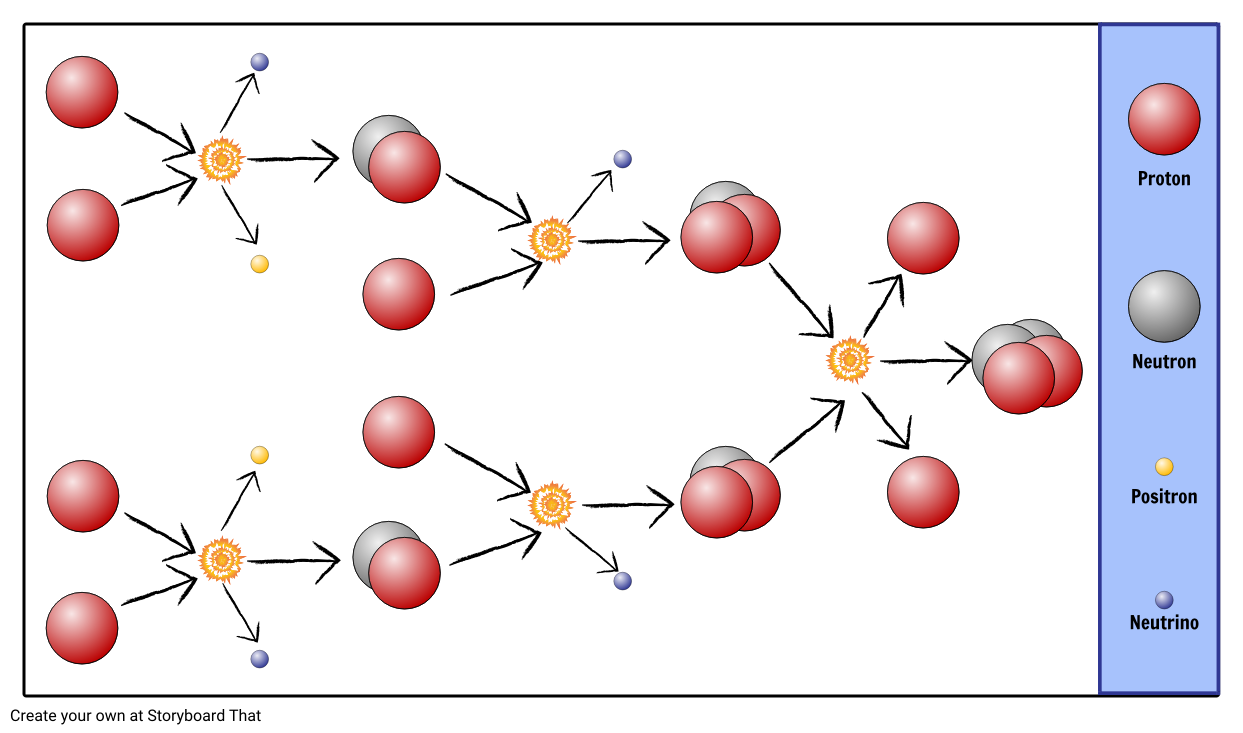
You could let students feel the air as it is released from the tyre - it should be colder than the ambient air as it is rapidly expanding as it escapes from the tyre. There is then less thermal energy and so the temperature decreases. As you deflate the tyre, you allow the gas to expand, the molecules are more spread out. As more particles are contained in the same volume, the air's temperature in the tyre increases. As more molecules are forced in by the pump, the air in the tyre is compressed and the total thermal energy increases because there are more molecules colliding inside the tyre. Each molecule has a certain amount of kinetic energy. So you are compressing or squeezing the gas. In the case of inflating a tyre, you are forcing more and more molecules into a given volume (assuming that the tyre is now at full capacity). They will find that the pump and tyre get hot! You could demonstrate this in class by getting learners to slightly over-inflate a tyre. If they are unfamiliar with this concept a good analogy is to think about over-inflating a bicycle tyre (without bursting it). In order to understand how collapsing gas clouds heat up to eventually form stars, learners need to understand that compressing a gas heats it up and that allowing a gas to expand cools it down. 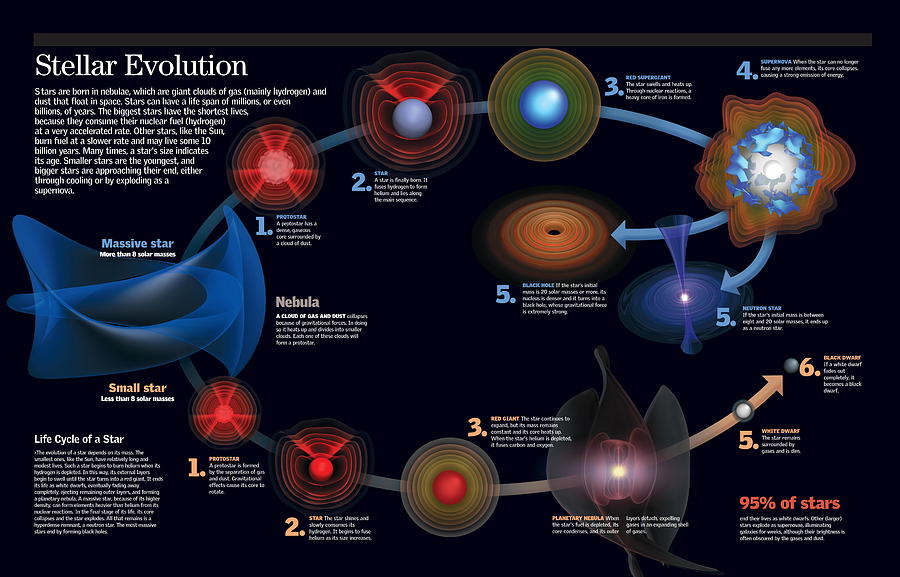
In this section learners will discover that stars are born in giant clouds of dust and gas, called nebulae, in space.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
